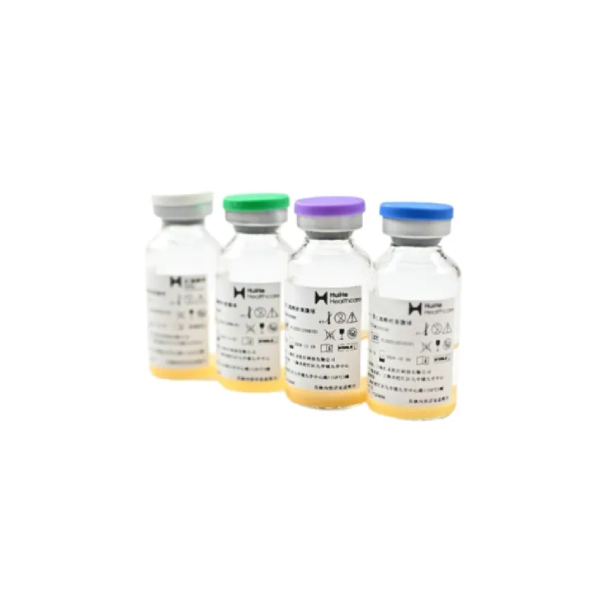
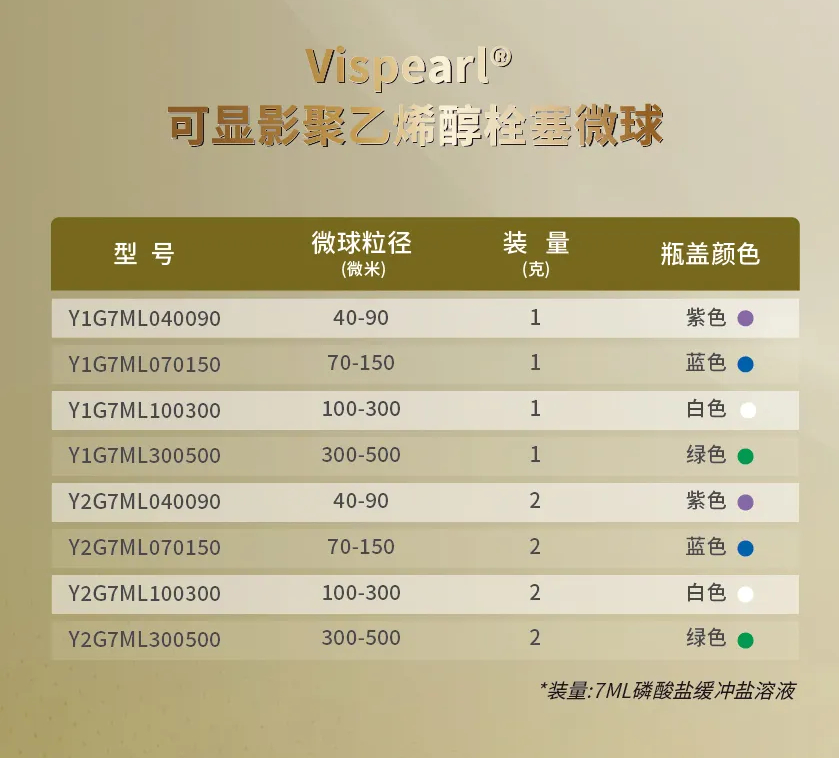
Vispearl® PVA Embolic Microspheres
Product Introduction
On March 25, 2024, H&H Medical's Vispearl®, the world's first fully visible drug-eluting microsphere in all sizes, received marketing approval (National Medical Device Approval No. 20243130560) from the National Medical Products Administration (NMPA). Vispearl®, the world's first fully visible drug-eluting microsphere in all sizes, demonstrates the R&D potential of China's medical industry and marks another solid step forward in the "Healthy China 2030" initiative. With its highly competitive pricing and exceptional quality, Vispearl® will provide the optimal treatment choice for tumor intervention. Vispearl® visible drug-eluting microspheres are indicated for the embolization of hypervascular solid organ malignancies.

In recent years, with the gradual popularity of the concept of fine embolization, the use of small particle size and ultra-small particle size embolization agents is also increasing. However, the only embolic agent that can achieve this particle size requirement in clinical practice is the traditional iodized oil emulsion. At the same time, iodized oil emulsion also has various problems, such as poor stability, insufficient density of embolization, not permanent embolization, serious postoperative adverse reactions and so on. However, the size of the currently commonly used microsphere embolization agents cannot meet the requirements of peripheral embolization.

To better address the aforementioned clinical challenges, H&H Medical developed Vispearl® radiopaque drug-eluting microspheres. Building on traditional drug-eluting microspheres, this globally unique, fully size-calibrated radiopaque microsphere is produced using patented iodinated radiopaque molecules (triiodobenzoic acid) combined with a critical-phase interfacial crosslinking process. It simultaneously resolves the limitations of conventional microspheres, such as excessive particle size preventing distal embolization and the inability to visualize post-procedurally. These optimizations enable the concept of precision embolization to better benefit a wide range of patients.
H&H Medical will hold a trial program of developing microspheres in the near future. Interested doctors can contact us backstage to communicate the specific trial process.
Product Feedback

Director of the Department of Interventional Radiology, Professor, and Doctoral Supervisor at Zhongshan Hospital Affiliated to Fudan University;
Deputy Director of the National Clinical Research Center for Radiology and Therapy
Vispearl®Developable Microspheres
warm congratulations to Vispearl®developable drug loaded microspheres officially listed!
the minimum size of this microsphere is 40 microns, which meets the clinical needs of our end "precision embolization. Advances in embolic agents and microcatheters have allowed our generation of interventional physicians to experience an era of rapid advances in TACE technology, and precise embolization can greatly reduce the impact on normal tissues.well known that there are no safe embolic agents, only those that are safe to use.40 μm particle size of the embolic agent for further efficacy escort, we can take the TACE treatment of liver cancer to a new height.

H&H Medical Biochemistry Research and Development Department
is honored to Vispearl®can be recognized by the State Drug Administration and many experts!
Vispearl®is the abbreviation of Visible Pearl. It is a developable drug-loaded microsphere with completely independent intellectual property rights and the first in China.
a quality pearl; more than three years have passed since I led the team to develop Vispearl®. We firmly believe that this "pearl" of Huihe can bring more certainty and better curative effect to doctors and patients. In order to develop more useful and economical medical devices, our footsteps have never stopped.

Academician of the Chinese Academy of Sciences, Professor, Doctor, Doctoral Supervisor,
.
warmly congratulate the first domestic Vispearl of developable drug-loaded microspheres.®officially listed!
TACE therapy is the recommended therapy for HCC treatment guidelines. We are in an era of technological explosion, and interventional therapy for liver cancer has undergone several generations of technological changes. From the earliest arterial infusion chemotherapy, to the use of lipiodol embolization, and then to drug-loaded microsphere embolization and radiation microsphere embolization.are very happy today that we have ushered in developable drug-loaded microspheres, and liver cancer embolization has entered the developable stage, which will greatly improve the safety and effectiveness of TACE treatment. I look forward to Vispearl®after the listing of all aspects of the performance.